Welcome to the August 18, 2017, edition of Pharma Industry News Update (aka PinUp). View the Web version of this issue here: http://bit.ly/PINUP081817
**********SUMMARY***********
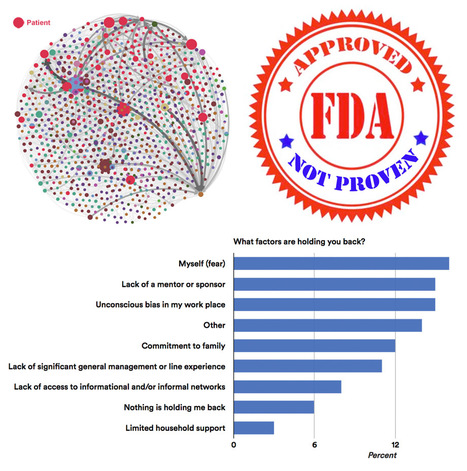
Is it a good idea to invite patients into the official social media discussion channels of major medical meetings? While results of a study summarized in this issue seem to support the usefulness of patients in helping disseminate scientific information from meetings, some experts believe opening discussions up to patients invites "trolling" by malicious agents.
Articles in this issue:
- “Patient Participation in Health Care Conference Social Media Discussions”; http://sco.lt/547M9J
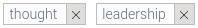
- “50% of Fast-Track Approved Drugs Have Not Been Proved Effective After 3 Years on the Market!”; http://sco.lt/8qhCzZ
- “Tumblr Low on Pharma's Most Popular Social Media Platforms List”; http://sco.lt/4wAMef
Further Reading:
- The Patients Included Movement: http://bit.ly/1FCbDDc
- GAO Report: FDA Expedites Drug Approvals, But Its Postapproval Oversight Stinks!: http://sco.lt/89j3Zp
- FDA is Lax in Enforcing Law Regarding Prescription Drug Postmarketing Studies: http://bit.ly/1PoqAsY
- Pfizer on Tumblr: http://sco.lt/6f2ecz
About Pharma Industry News Update
The Pharma Industry News Update (aka PinUp) is published every Tuesday and Friday as part of the Pharma Marketing News subscription service. It features curated pharma industry news and views of topical interest from a variety of sources. If you'd like to receive this newsletter, subscribe here.
Via Pharma Guy



 Your new post is loading...
Your new post is loading...