Journal of Medical Internet Research - International Scientific Journal for Medical Research, Information and Communication on the Internet
Get Started for FREE
Sign up with Facebook Sign up with X
I don't have a Facebook or a X account


|
PATIENT EMPOWERMENT & E-PATIENT
Patients as the healthcare system "missing link" #digitalhealth #hcsmeu #hcsm #epatient Curated by Lionel Reichardt / le Pharmageek |
 Your new post is loading...
Your new post is loading...

|
Scooped by Lionel Reichardt / le Pharmageek |
Encouraging patients to help write and add notes to their personal medical charts -- a task typically handled only by medical professionals -- may help patients feel more involved with their own care and improve relationships with their doctors, a new study has found.

Según un estudio publicado en Annals of International Medicine, una investigación elaborada por investigadores de UCLA Health y Beth Israel Deaconess Medical Center, ambos de EEUU, señala que la participación de los pacientes agregando su propia información en la Historia Clínica Electrónica o HCE (lo que denominan sus propias notas médicas) mejora su involucración en su propia atención médica y mejora la relación del paciente con los profesionales médicos. Además facilita el trabajo a los propios médicos en cuanto a la documentación de la HCE, ya que gracias a esto invertirán menos tiempo. En ésta los propios pacientes añaden información como síntomas o problemas médicos que han ido presentando desde su última visita médica, así como objetivos para próximas sesiones. Esto permitiría por lo tanto a los médicos centrarse más en la atención al paciente. Está previsto que en 2018 comiencen los primeros programas piloto en algunos centros médicos en EEUU.

|
Rescooped by Lionel Reichardt / le Pharmageek from Pharmaguy's Insights Into Drug Industry News |
At a time when drug company lobbyists are widely vilified as icons of avarice, patient advocacy groups still wear the white hats.
But those organizations, which promote cures for every type of cancer and hundreds more diseases, have come under criticism lately for favoring their drug company funders in contests on Capitol Hill.
In one case, a diabetes group accepted money from food companies and played down the health risks from their high-sugar products; in another case, a mental health association, reliant on drug company dollars, opted to keep quiet about the soaring prices of its antidepressants. And many of the patient advocacy groups pushing for passage of the 21st Century Cures Act, which consumer groups argue rolls back patient protection, are funded in large part by pharmaceutical firms.
“The public should be concerned about this for many reasons,” said Jonathan H. Marks, director of the bioethics program at Pennsylvania State University. “One of the most important is that patient advocacy groups have credibility with policymakers — as corporate donors are well aware. Policymakers tend to assume that these organizations are acting in the interests of patients, or public health more broadly.”
But, said Marks, this is not always the case when the groups are reliant on drug or device industry donations — a point not often discussed when they lobby the Food and Drug Administration to speed new drugs to market, participate in National Institutes of Health panels, bring patients to testify before Congress, or advise patients on courses of treatment.
A study published Tuesday in JAMA Internal Medicine offers evidence of the ways in which patient advocacy organizations, or PAOs, rely on industry dollars.
The research, led by Susannah Rose of the Cleveland Clinic, shows that PAOs receive industry funding more often than previously believed. Some of the groups’ leaders, responding to a confidential survey, also acknowledged donor pressure to take policy positions that are best for the donors. Others said they doubted their own level of independence.
Rose, director of research in the Cleveland Clinic Office of Patient Experience, and her colleagues surveyed 439 patient advocacy organizations across the United States.
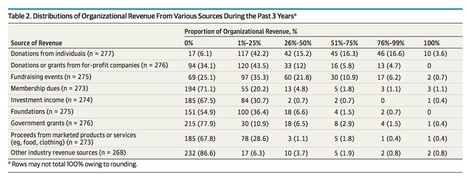
Of the 289 groups that responded, more than two-thirds reported receiving industry funding, with a median of $299,000. Twelve percent said they received over half their funding from industry. Almost 9 percent received $1 million or more. The pharmaceutical, medical device, and biotechnology sector accounted for a median of 45 percent of the donations. Only one quarter of the groups said they had policies on disclosing their financial relationships.
Further Reading:
- Transparency is Good in Theory, But Not in Practice; http://sco.lt/6qCqTR
- #Pharma to Patient Advocacy Groups Questioning High Drug Prices: "Why Are You Doing This to Us?"; http://sco.lt/4sOB7J
- Holy Sh*t! Is There No End to Mylan's Shenanigans? Paying Off Patient Groups to Lobby!; http://sco.lt/6Sl0ld
- UK Patient Groups that Backed New Cancer Drug Received £ from #Pharma Firm; http://sco.lt/84W3En
- Majority of Patients’ Groups Siding With Pharma Against Medicare Part B Pricing Reforms Receive Industry Funding; http://sco.lt/574i6D
- #Pharma's "Patient Centricity" Pays Off: Patient Groups Mum on Drug Costs; http://sco.lt/8ydeuv
Via Pharma Guy

|
Rescooped by Lionel Reichardt / le Pharmageek from Pharma Industry Regulation |
Patients and their advocates are getting an ever-larger voice in how medical research is carried out. They participate in the design of experiments and have a greater say in what outcomes they care about most — and it's not always simply living longer.
There are now hundreds of patient groups, focusing on everything from arthritis to xeroderma pigmentosum. And patient participation in medical research is no longer an informal matter. The concept is written into federal laws, including one that funds the Food and Drug Administration. Since 2013, the FDA has held more than 20 meetings in which patients are invited to lay out what actually matters to them.
The FDA historically hasn't considered its work from the patient's point of view. It generally starts with a company pitching a new drug and is supposed to answer the narrower question: Is the product safe and effective? Now that the FDA knows what patients care about, it can suggest that drug companies measure those outcomes in the course of testing new products.
The FDA has already demonstrated that it's paying more attention to what patients want. Earlier this year, after hearing impassioned pleas from parents and their scientific allies, the agency temporarily approved a drug for Duchenne muscular dystrophy, even though the science in hand didn't provide proof positive that the drug was effective (read “FDA Succumbs to Industry-Sponsored "Patient Power" & Accelerates Approval of Sarepta's Duchenne Drug”; http://sco.lt/4mgzvV).
Patients considered that a victory. And the company could start selling its drug for $300,000 per patient per year.
"The bad news about what's happening is that so much of patient input is involving patients who are working with or who are recruited by drug companies," cautions Diana Zuckerman, who runs a consumer group called the National Center for Health Research.
These companies pour millions of dollars into disease advocacy organizations and provide information to patients. So the patient voices are sometimes inadvertently channeling commercial interests, Zuckerman says.
With that system, patients also hear more about potential benefits than risks, she says. Clearly patients should have a voice in how big a risk they are willing to take, she says, but within limits.
Via Pharma Guy

|
Rescooped by Lionel Reichardt / le Pharmageek from Pharma: Trends and Uses Of Mobile Apps and Digital Marketing |
A report published by PricewaterhouseCoopers’s Health Research Institute reveals that the FDA, which developed the Patient-Focused Drug Development program in 2012 to better engage with patients, has held 21 disease-specific meetings to gain insight from them.
So far the meetings have focused both on well-known diseases such as breast cancer and HIV, and not so well-understood maladies such as female sexual dysfunction and Chagas disease. But what questions is FDA asking of patients and how often do they recur in the 21 meetings held? (See chart here).
Here are some FDA actions that should warm the cockles of the heart of patient engagement advocates in the future, according to the report. These are based on FDA’s PDUFA (Prescription Drug User Fee Act) reauthorization proposal:
- Between 2017 and 2021, the agency expects to hire additional staff focused on engaging with patients and facilitating the development as well as use of patient-focused drug development methods.
- In 2018, FDA is expected to provide draft guidance describing approaches to collecting patient and caregiver input.
- In 2019, the agency plans to issue draft guidance describing how companies can collect information from patients, and how that information can be used in the drug development and regulatory decision-making process.
- Another draft guidance describing how meaningful patient perspectives and information can be collected in clinical trials is expected in 2020.
- And finally, a draft guidance on patient-reported outcome measures to replace the one released in 2009 is set to be available come 2021.
Draft guidance aside, FDA is also home to a treasure trove of data. And now the agency is opening up access so that third parties can turn that information into insight. For instance, through the openFDA database, companies like Iodine and Advera Health Analytics are turning adverse event information on drugs into a searchable format, the report found.
While FDA is dabbling with patient engagement strategies, on the flip side, patient advocacy groups are also getting involved in the regulatory space (read, for example, “How a #pharma Funded ‘Grassroots’ Patient Advocacy Campaign Changed FDA's Approval Process”; http://sco.lt/4tqlbF). The report mentions one — Parent Project Muscular Dystrophy — that developed a guidance document so that companies could use it to develop drugs for Duchenne Muscular Dystrophy. That guidance document ended up being modified and adopted by the FDA for use by companies developing drugs for the disease.
“While patient input is unlikely to improve the approval chances of a drug lacking solid efficacy and safety data, regulators may be more willing to work with companies that are developing a product in close concert with engaged or especially ill patient populations,” it advised.
Via Pharma Guy, Giuseppe Fattori, eMedToday


Meanwhile, “Patient Advocates - and Docs with Ties to Pharma - Turn Up the Heat on FDA”; http://sco.lt/8koMLZ

|
Rescooped by Lionel Reichardt / le Pharmageek from Pharmaguy's Insights Into Drug Industry News |
An independent study by PatientView
Findings based on a survey of 1,150 patient groups (from 58 countries and of differing specialties)Survey conducted mid-November 2014 to mid-January 2015Patient-group feedback on the corporate reputation of the entire pharma industry during 2014Patient-group feedback on the corporate reputation of 37 individual pharma companies in 2014Results for 2014 are compared with those of 2013, 2012, and 2011
The 37 pharma companies reviewed in the study are: AbbVie l Actavis l Allergan l Amgen l Astellas l AstraZeneca l Baxter International l Bayer l Biogen Idec l Boehringer-Ingelheim l Bristol-Myers Squibb l Celgene l Eisai l Eli Lilly (Lilly) l Gilead l GlaxoSmithKline (GSK) l Grũnenthal l Ipsen l Janssen l Lundbeck l Menarini l Merck & Co (USA) l Merck KgA (Germany) l Mylan l Novartis l Novo Nordisk l Otsuka l Pfizer l Roche l Sanofi l Servier l Shire l Stada Arzneimittel l Takeda l Teva l UCB l ViiV Healthcare
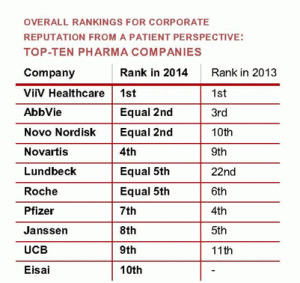
The corporate reputation of individual pharma companies (as seen from a patient perspective) shows some significant changes between 2014 and 2013 (37 companies assessed in 2014; 33 companies in 2013).
Via Pharma Guy

Several of the patient organizations participating in this survey receive funding from the pharmaceutical industry. This is common practice especially in the EU where pharma companies cannot promote product directly to consumers but must depend upon intermediaries such as patient organizations -- some of which pharma companies set up themselves -- to help them reach consumers.
Also note that Lundbeck (after slumping to 22nd in 2013—almost certainly because patient groups reacted to the June 2013 news that the company had been fined by the European Commission) has managed to move back up the rankings in 2014 to 5th. How quickly patients forget! It's difficult to believe. More likely is that Lundbeck spread goodwill via increased grants to patient groups. I'm just speculating here.

In response to Pharmaguy's 'speculation'. There is a close relationship between pharma companies and patient groups but not for the reasons he cited and certainly not to the point that it would bias the outcome.

|
Rescooped by Lionel Reichardt / le Pharmageek from healthcare technology |
These are the slides from my talk at the 4th Annual Putting Patients First Conference in Mumbai.
If god were to manifest the world using technology, he would first create something like social media. Conceptually provide technology with the ability to understand the thoughts of a population
SocMed leaves behind the old model of 1-to-1 communication – “talking to someone over the phone” Enables one-to-many communication (via blogs or microblogging) or many-to-many communication (discussion forums, social walls). Now anyone can setup an online community site/portal to represent a small or big offline community.
Further, anyone can setup an online site related to a treatment, a disease, a doctor, a drug , a concept or anything and see it grow into a popular site which in effect is simply the manifestation of a community which exists/ed but which no one ever knew of.
Via nrip

Great info! I think SocMed will boost the thought of population and will effect change in local communities.